Welcome to the “52 in 52” series! This collection of posts features recently published must-know articles. For our first post, Brannon Inman looks at the HALT-IT trial.

Authors: Brannon Inman (Chief Resident, Emergency Medicine Physician, San Antonio, TX) // Reviewed by: Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Effects of a high-dose 24-h infusion of tranexamic acid on death and thromboembolic events in patients with acute gastrointestinal bleeding (HALT-IT): an international randomized, double-blind, placebo-controlled trial
AKA: The “HALT-IT” Trial
Clinical question:
Does TXA reduce bleeding-related death in patients with GI bleed?
Study design:
- Block randomized placebo-controlled trial

PICO:
Population:
- Included patents in 15 countries:
- UK, Pakistan, Nigeria, Egypt, Malaysia, Georgia, Romania, Nepal, Sudan, Saudi Arabia, Spain, Ireland, Albania, Papua New Guinea, and Australia
- Included patients above age of adulthood for respective countries (16 or 18 depending on country) with gastrointestinal bleed (upper or lower GI bleed)
- Clinician caring for patient must have been uncertain if TXA would be helpful
- Diagnosis of bleeding needed to be significant, and clinical, defined as “At risk for bleeding to death”. Authors included patients with:
- Tachycardia
- Hypotension
- Shock
- Expected need for endoscopy/surgery
- Exclusion criteria not stated
Intervention:
- 1g TXA load (in 100 cc 0.9% NS administered over 10 min)
- Following load, 3g TXA infusion administered over 24 hours
Comparator:
- A placebo solution and equal volume of isotonic crystalloid administered over 24 hours
Randomization:
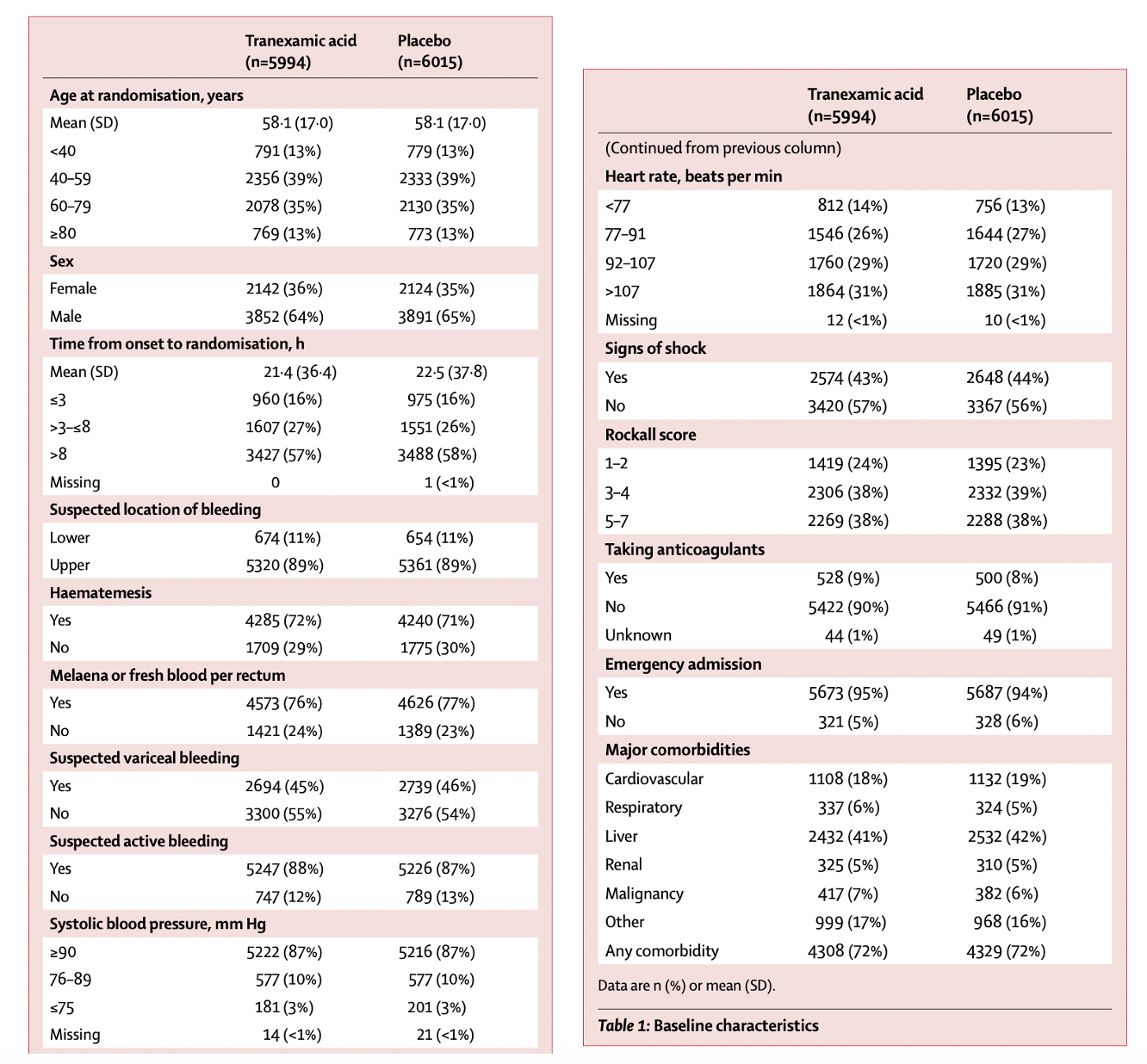
Treatment arms were fairly well balanced.
Outcome:
- Primary outcome included death due to bleeding within 5 days of randomization
- Authors found no significant difference in primary outcome (death due to bleeding at 5 days) (RR 0.99, 95% CI 0.82-1.18)
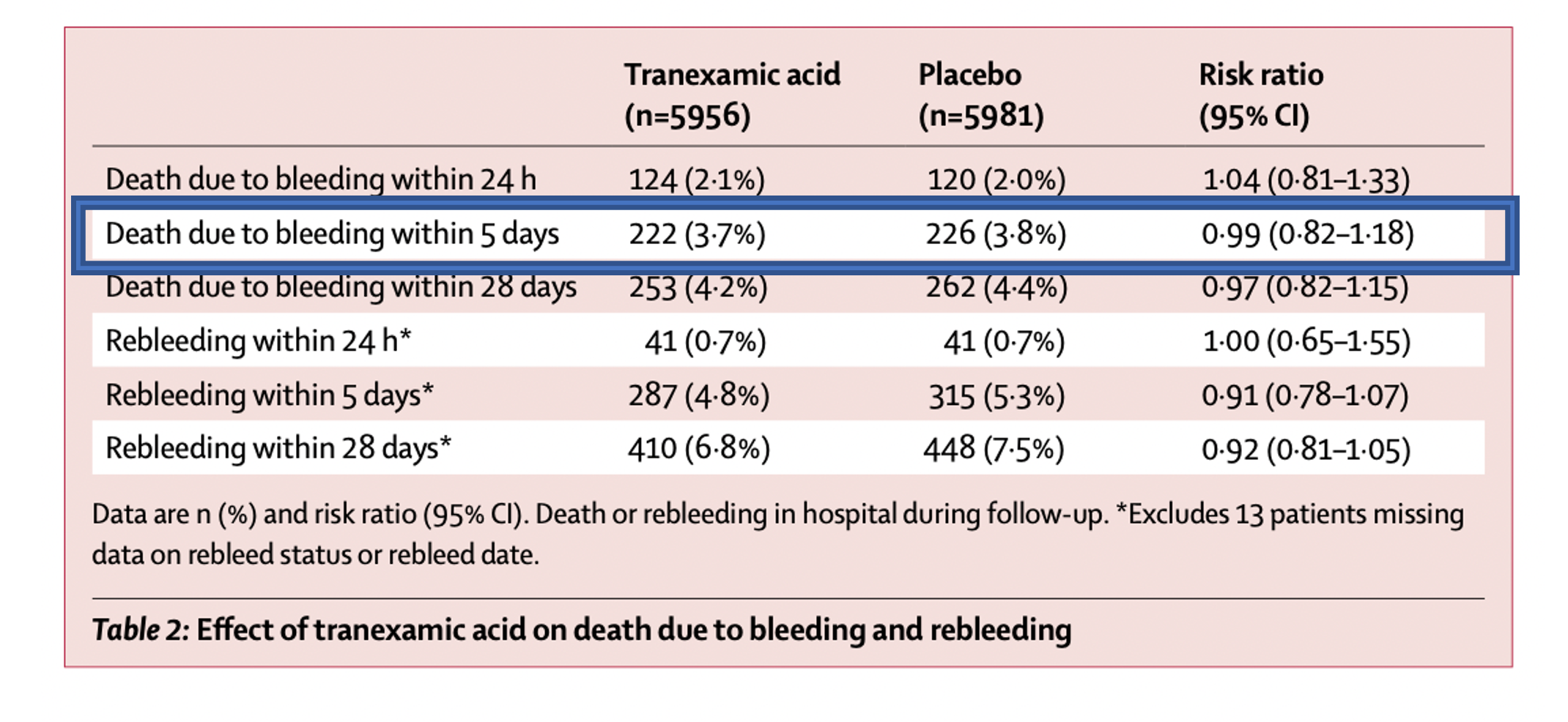
- Statistically significant risk observed for occurrence of venous thromboembolic events (PE and DVT) and seizures
- VTE RR 1.85, 95% CI 1.15-2.98, NNH 250
- No increase in arterial thromboembolic events (MI and stroke)

Take away:
- This is a negative trial.
- Included patients with upper and lower GI bleeds as one group, which is not how we typically classify these patients. The majority of patients had variceal bleed, accounting for 75% of deaths. Less than 10% of included patients were on anticoagulants.
- Only 69% of patients required transfusion, and 87% had SBP > 90 mm Hg with 30% having a HR > 107.
- Time from onset to randomization < 3 hours was present in 16% of patients. Most patients received TXA over 8 hours from time of bleeding onset.
- Primary outcome changed from all-cause mortality to GI bleeding related mortality in 5 days. Reportedly this decision was made blind to the accumulating data. Changing a primary outcome from a patient-centered outcome to a disease-centered outcome is odd. Our patients do not care what they die from, nor do their families. With the used primary outcome, patients could have been dying from TXA complications, and this would not have been captured (albeit this is unlikely).
- Very few trials have shown harm associated with TXA. This trial showed a statically significant risk of VTE and seizures.
My take:
- This is a well-constructed, multicenter RCT comparing TXA versus placebo for patients with acute GI bleeding. Based on the results from this RCT, TXA does not reduce death from GI bleeding and may increase risk of VTE and seizure.
- I won’t be using TXA for GI bleed patients in my clinical practice.
Reference:
- Brenner A, Afolabi A, Ahmad SM, et al. Tranexamic acid for acute gastrointestinal bleeding (the HALT-IT trial): statistical analysis plan for an international, randomised, double-blind, placebo-controlled trial. Trials. 2019;20(1):467. Published 2019 Jul 30. doi:10.1186/s13063-019-3561-7






