Author: Thijs Wolf, DO (EM Resident Physician, University of Rochester Medical Center); Andrew Bragg, MD (EM Attending Physician, University of Rochester Medical Center) // Reviewed by: Sophia Görgens, MD (EM Physician, Northwell, NY); Cassandra Mackey, MD (Assistant Professor of Emergency Medicine, UMass Chan Medical School); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Welcome to EM@3AM, an emDOCs series designed to foster your working knowledge by providing an expedited review of clinical basics. We’ll keep it short, while you keep that EM brain sharp.
A 36-year-old female presents to the ED after experiencing an episode of sudden syncope. The patient has no significant past medical history, and reports that she was walking to the end of her driveway to retrieve her mail when she suddenly collapsed to the ground. She recalls waking up on her driveway with concerned neighbors gathered around her. The patient states that this has never happened before. On arrival, vital signs are stable with a HR of 71, BP of 121/71 mmHg, RR of 17 bpm, and SpO2 of 97% on room air. Physical exam reveals a well-appearing female in no acute distress. Cardiac exam is unremarkable with normal S1/S2 without rubs, gallops or murmurs. Neurological exam is also unremarkable. Laboratory testing shows normal cardiac markers and no signs of inflammation. Her presenting EKG is shown below.
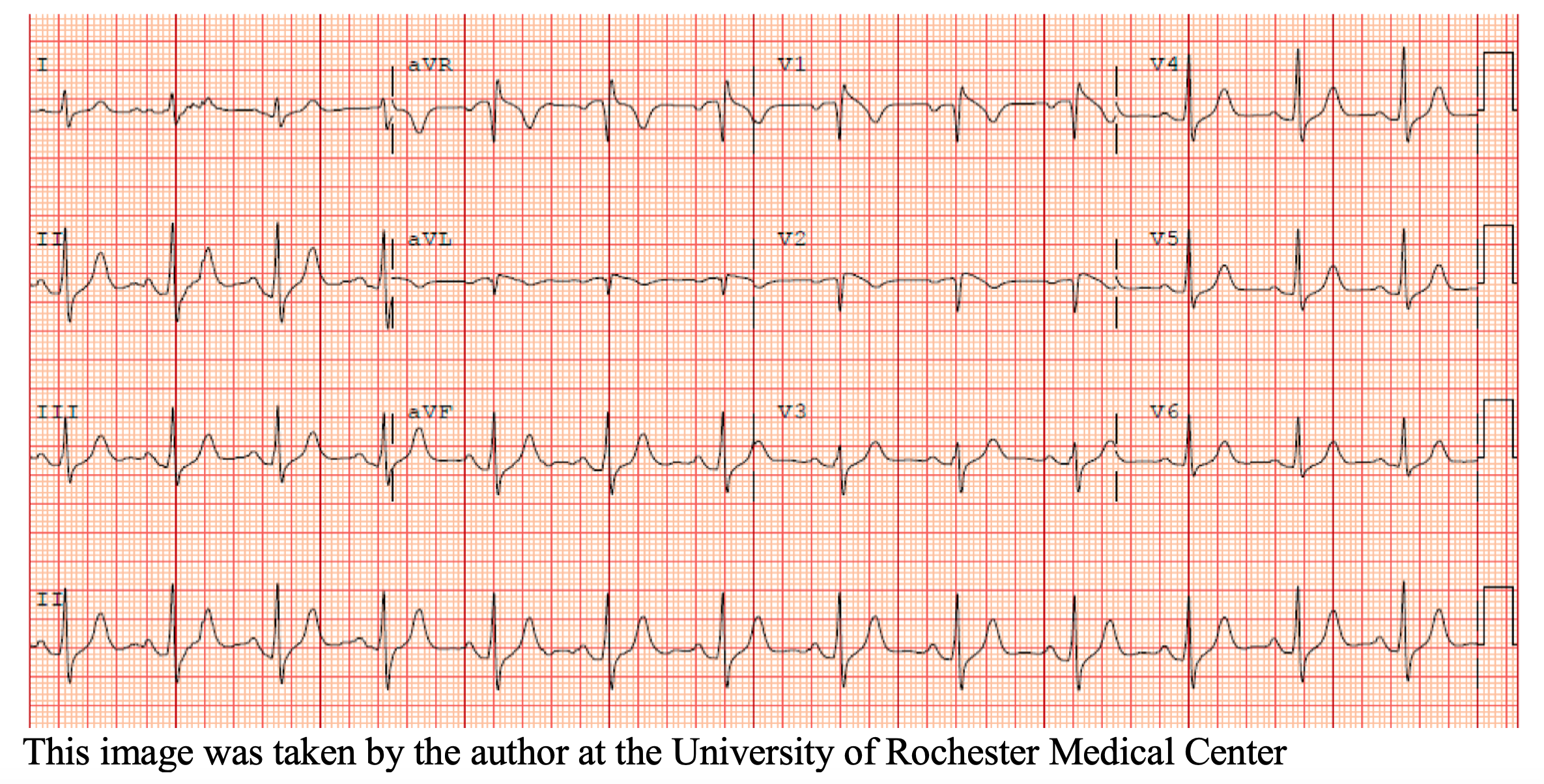
What is the most likely diagnosis, and what are the next steps in management?
Answer: Brugada Syndrome
Epidemiology and Etiology
- Rare genetic disorder that affects approximately 1 in 5,000 individuals worldwide1.
- More common in males than females, with a male-to-female ratio of approximately 8:11.
- Most commonly found in individuals of Southeast Asian descent, with a prevalence of up to 5% in some populations3.
- Caused by mutations in the SCN5A gene, which encodes the alpha subunit of the cardiac sodium channel4. Other genes, such as SCN1B, have also been implicated in the disorder5.
- The exact mechanisms by which these genetic mutations lead to the characteristic ECG and arrhythmias seen in Brugada Syndrome are not yet fully understood, but they are thought to involve abnormalities in cardiac ion channels that affect the flow of sodium and calcium ions across cardiac cell membranes6.
Clinical features
- Patients often present after an episode of sudden syncope, although Brugada syndrome can also be found on a routine EKG.
- May experience syncope or sudden cardiac arrest due to the development of ventricular arrhythmias such as ventricular fibrillation1.
- Presentation can be triggered by factors such as:
- Fever
- Alcohol consumption
- Cocaine7
- Certain medications such as sodium channel blockers and tricyclic antidepressants8.
- May have other associated clinical features such as chest pain, palpitations, and shortness of breath1.
Evaluation
- Obtain a detailed family history of sudden cardiac death, syncope, or known Brugada Syndrome.
- ECG to evaluate for arrhythmia.
- Evaluate for any other cardiac abnormalities or comorbidities that may increase the risk of arrhythmias, such as electrolyte imbalances, myocardial ischemia, structural heart disease and infection.
- The routine assessment of cardiac biomarkers such as troponin is not recommended in patients with suspected Brugada syndrome9.
Diagnostic criteria
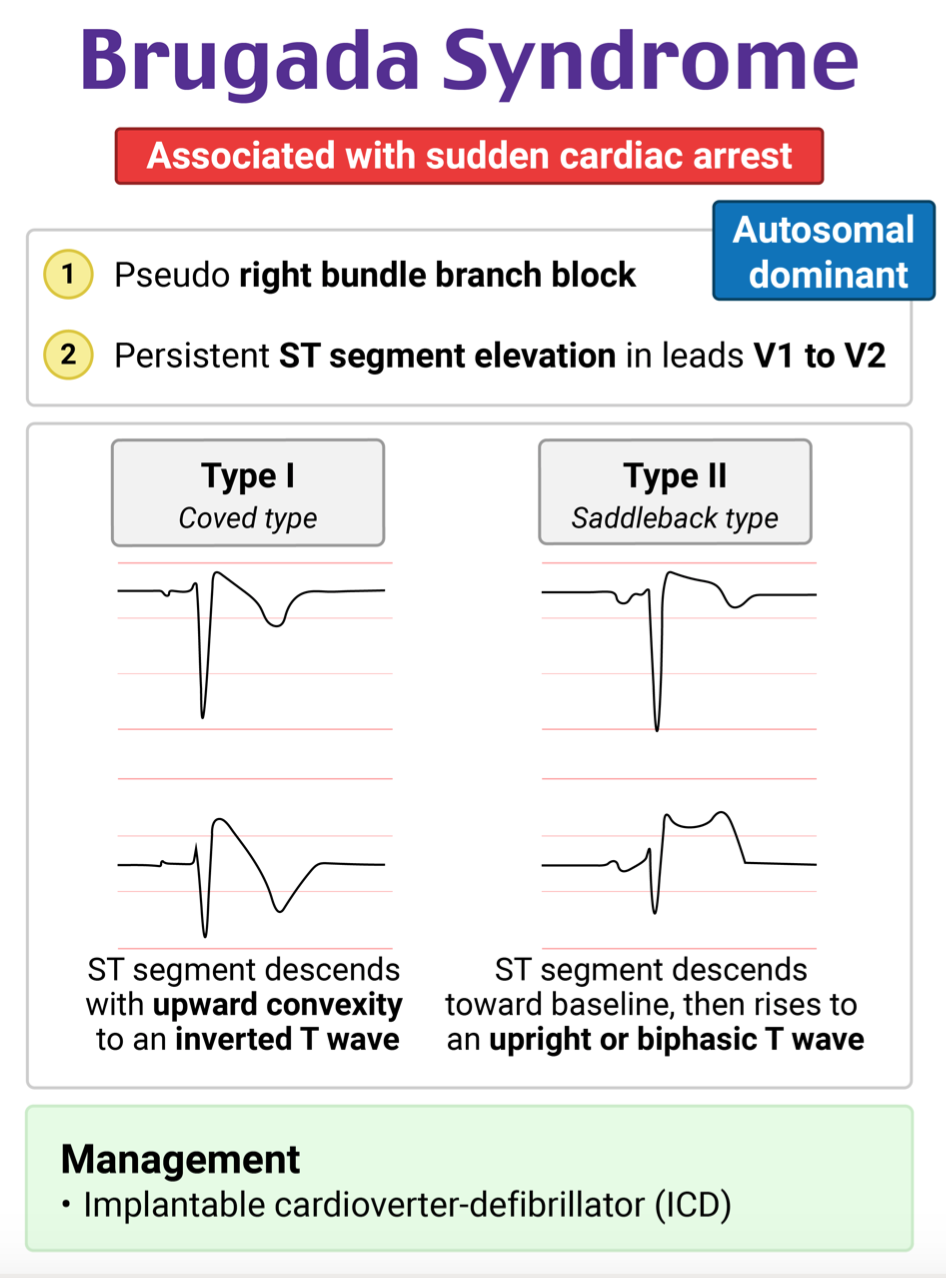
There are 2 types of ECG patterns that are part of the diagnostic criteria of Brugada syndrome
- Type 3 is described below and was previously part of the diagnostic criteria but is now believed to be a normal variant.
- Type 1
- “Brugada sign”
- The only type that is considered diagnostic.
- Requires the classic ECG pattern + diagnostic criteria.
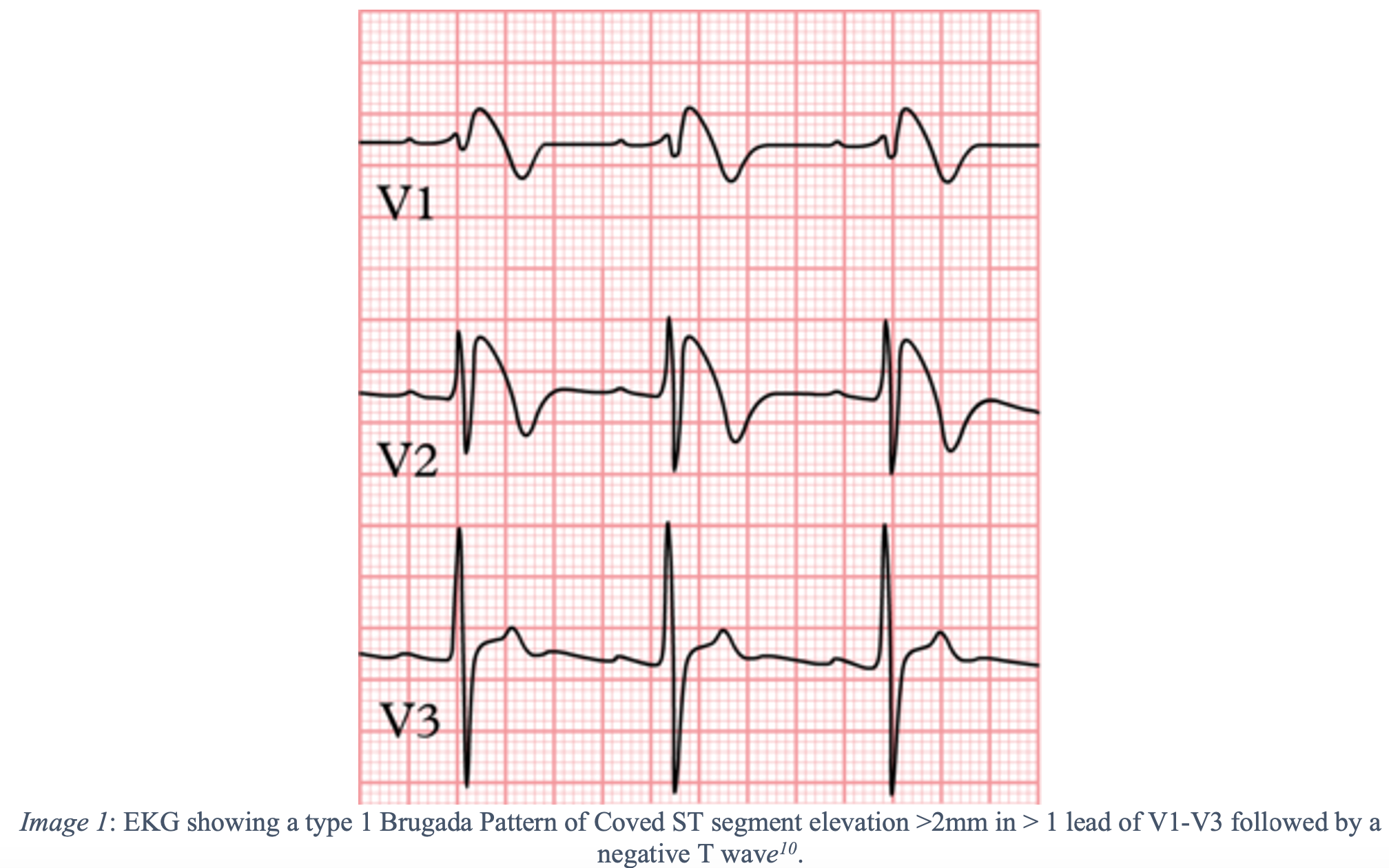
- The presence of a type 1 Brugada ECG pattern – Coved ST segment elevation >2mm in > 1 lead of V1-V3 followed by a negative T wave in conjunction with one or more of the following criteria:
- Documented ventricular fibrillation or polymorphic ventricular tachycardia
- A family history of sudden cardiac death at <45 years of age or syncope of undetermined origin at any age
- History of a type 1 ECG pattern in an immediate family member.
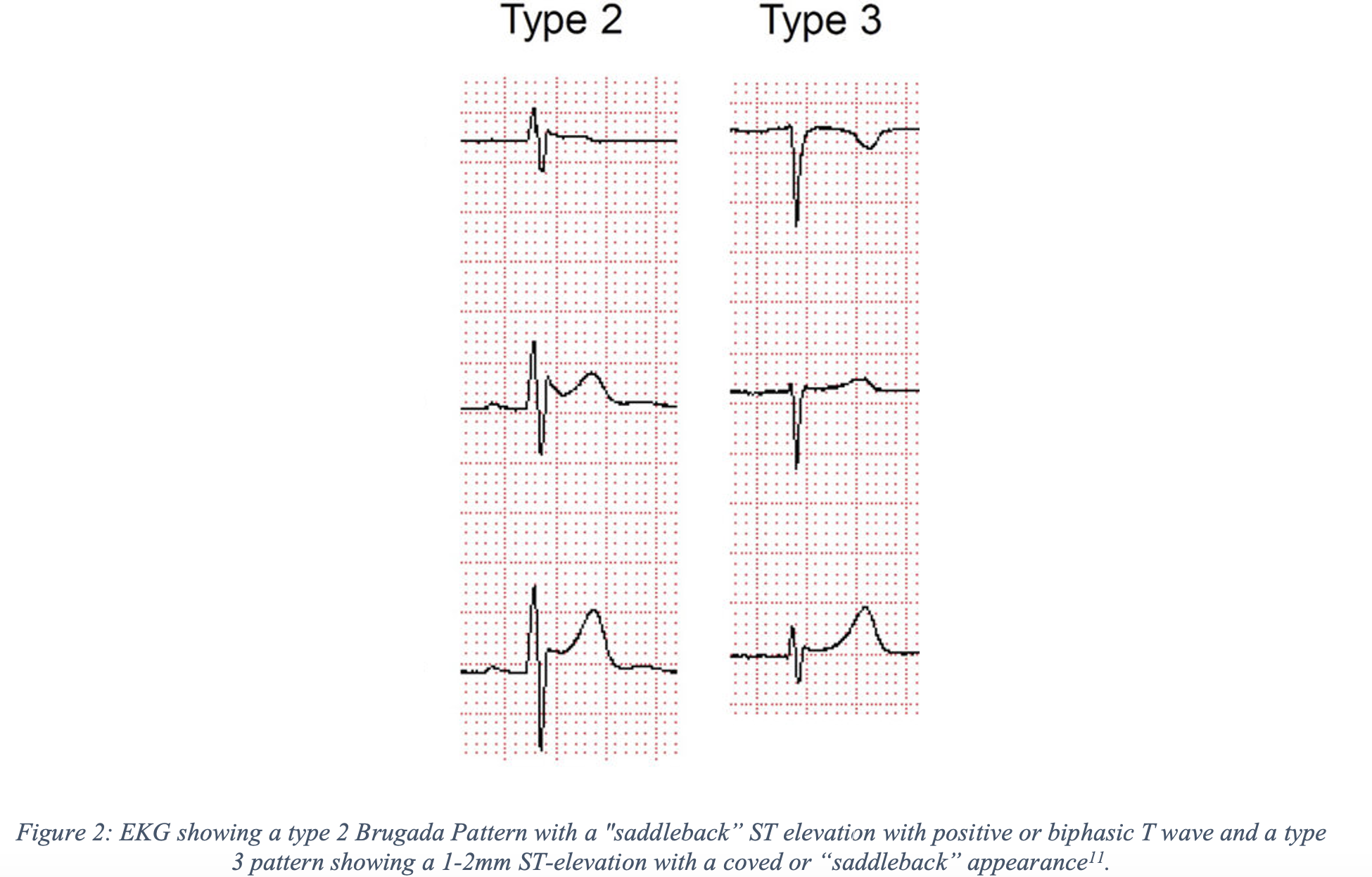
- Type 2
- “Saddleback” ST elevation with positive or biphasic T wave.
- Suggests the possibility of Brugada Syndrome, and further testing is necessary for diagnosis.
- Type 3
- 1-2mm ST-elevation with a coved or saddle-back appearance.
- Previously thought to be suggestive of Brugada syndrome but now is considered a normal variant.
Management
- All symptomatic patients with a type 1 pattern should be admitted for EP consultation.
- Asymptomatic patients with a type 1 pattern or patients with type 2 pattern may be considered for outpatient cardiology and EP evaluation, although this remains controversial.
- The only proven therapy is an ICD.
- In patients with Brugada syndrome who have survived a life-threatening arrhythmic event, long-term use of quinidine has been shown to decrease recurrent arrhythmic events12.
- 15% of patients on Quinidine therapy still experienced a life-threatening arrhythmia suggesting it cannot be used as an alternative to an ICD12.
Pearls
- Important cause of sudden cardiac death, especially in young, apparently healthy individuals.
- AICD is the mainstay of therapy for patients with Brugada syndrome who have had cardiac arrest or who have other high-risk features.
- Admit symptomatic patients to the hospital for further evaluation and EP studies.
- Patients should be counseled to avoid triggers such as fever, alcohol, and certain medications that may precipitate arrhythmias.

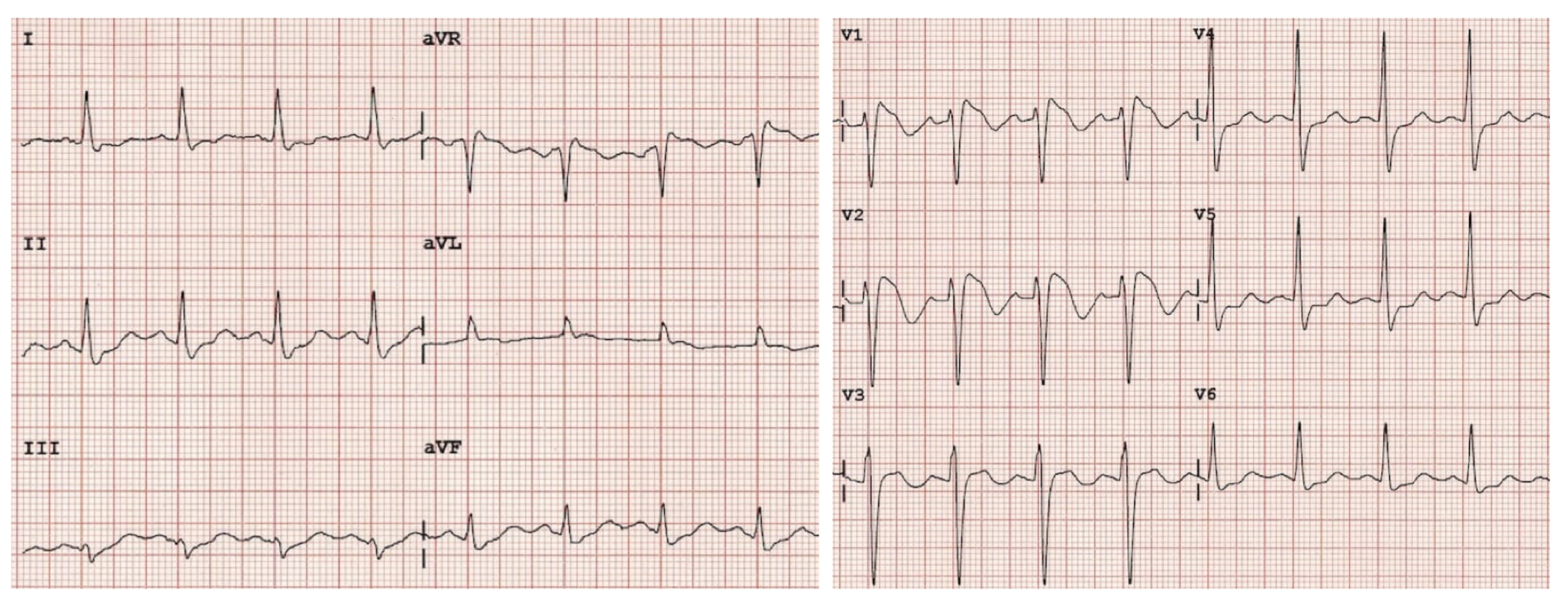
A 23-year-old man with no significant medical history presents to the emergency department for evaluation of a syncopal episode. The patient reports that he was doing light housework when he suddenly passed out. He had no preceding symptoms and now feels completely back to normal. He reports a family history of a cousin who died at age 41 from a “heart problem,” but he does not know any further details. His HR is 85 bpm, and his BP is 125/74 mm Hg. His physical exam is normal, with no cardiac murmur on auscultation. The patient’s ECG is shown above. A troponin-I level is < 0.04 ng/mL. Which of the following is the most likely cause of the findings seen on ECG?
A) Cardiac ischemia
B) Elevated serum potassium level
C) Genetic disorder of the myocardium
D) Sodium channelopathy
Answer: D
Brugada syndrome is a genetic disorder resulting in the loss of function of the cardiac sodium channel, which increases the risk of spontaneous ventricular tachycardia and ventricular fibrillation. Male patients are most commonly affected, particularly those of Southeast Asian heritage, where the incidence of the disease is higher. An ECG will show a pseudo-right bundle branch block(RBBB) in leads V1–V2 with ST elevation from the J-point. The type I pattern, as seen in the ECG above, has coved ST segment elevation > 2 mm in more than one lead (V1–V3) that terminates in a negative T wave. A type II pattern demonstrates ST elevation that is concave upward in a saddleback pattern, while a type III pattern has ST segment elevation of < 1 mm in a coved or saddleback pattern. Types II and III are less sensitive and specific for Brugada syndrome than type I. Brugada sign refers to the findings on ECG, while Brugada syndrome is the combination of ECG findings with one of the following: documented ventricular tachycardia or fibrillation, syncope, family history of sudden cardiac death at < 45 years old, or inducible ventricular tachycardia with programmed electrical stimulation. Patients with Brugada syndrome should have a prompt cardiology consultation for consideration of implantable defibrillator placement due to the high risk of sudden cardiac death.
While the patient does have ST segment elevation, the pattern and history are consistent with Brugada syndrome rather than cardiac ischemia (A). Patients with Brugada syndrome should test negative for cardiac enzymes, given that the ECG changes can mimic an ST elevation myocardial infarction. Hyperkalemia (B) can cause ECG findings of peaked T waves, bradydysrhythmias, conduction blocks, and QRS widening. Patients with Brugada syndrome may have an RBBB pattern but will have normal serum potassium levels and no other expected ECG findings of hyperkalemia. There is nothing in this patient’s history to suggest hyperkalemia as the cause of his syncope, and his ECG demonstrates a classic type I Brugada sign. A genetic disorder of the myocardium (C) may refer to an underlying diagnosis of hypertrophic cardiomyopathy (HCM) or arrhythmogenic right ventricular cardiomyopathy (ARVC), both of which are common causes of sudden cardiac death in young people. In HCM, an ECG may demonstrate increased voltages in the precordial leads and deep, narrow Q waves in the lateral leads, I, aVL, and V5–V6. Patients with HCM will also exhibit a high-pitched midsystolic murmur best heard at the left lower sternal border. The ECG in patients with ARVC may show T wave inversion in V1–V3 in the absence of an RBBB or a small upward deflection at the end of the QRS complex in V1, referred to as an Epsilon wave.
The demographic information in the above explanation follows AMA Manual of Style terminology and may not match the language used in the references. Race is a social construct that is often correlated to certain medical conditions in the literature and evidence-based guidelines. Our hope is to inspire a change in the way race is used in the medical community.
References
- Antzelevitch C, Brugada P, Borggrefe M, et al. Brugada syndrome: report of the second consensus conference: endorsed by the Heart Rhythm Society and the European Heart Rhythm Association. Circulation. 2005;111(5):659-670.
- Mizusawa Y, Wilde AA. Brugada syndrome. Circ Arrhythm Electrophysiol. 2012;5(3):606-616.
- Priori SG, Napolitano C, Gasparini M, et al. Natural history of Brugada syndrome: insights for risk stratification and management. Circulation. 2002;105(11):1342-1347.
- Chen Q, Kirsch GE, Zhang D, et al. Genetic basis and molecular mechanism for idiopathic ventricular fibrillation. Nature. 1998;392(6673):293-296.
- Wilde AA, Antzelevitch C, Borggrefe M, et al. Proposed diagnostic criteria for the Brugada syndrome: consensus report. Circulation. 2002;106(19):2514-2519.
- Dumaine R, Towbin JA, Brugada P, et al. Ionic mechanisms responsible for the electrocardiographic phenotype of the Brugada syndrome are temperature dependent. Circ Res. 1999;85(9):803-809.
- Littmann L, Monroe MH, Svenson RH. Brugada-type electrocardiographic pattern induced by cocaine. Mayo Clin Proc2000;2013:845.
- Priori SG, Wilde AA, Horie M, et al. HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes: document endorsed by HRS, EHRA, and APHRS in May 2013 and by ACCF, AHA, PACES, and AEPC in June 2013. Heart Rhythm. 2013;10(12):1932-1963.
- Frustaci A, Priori SG, Pieroni M, Chimenti C, Napolitano C, Rivolta I, Sanna T, Bellocci F, Russo MA. Cardiac Histological Substrate in Patients With Clinical Phenotype of Brugada Syndrome. Circulation. 2005;112(22):3680-3687. doi: 10.1161/CIRCULATIONAHA.105.539307.
- Postema PG. Brugada Syndrome. ECGpedia, 22 May 2013, https://en.ecgpedia.org/wiki/Brugada_Syndrome.
- Take Y, Morita H, Toh N, Nishii N, Nagase S, Nakamura K, Kusano KF, Ohe T, Ito H. Identification of high-risk syncope related to ventricular fibrillation in patients with Brugada syndrome. Heart Rhythm. 2012 May;9(5):752-9.
- Mazzanti A, Tenuta E, Marino M, et al. Efficacy and limitations of quinidine in patients with Brugada syndrome. Heart Rhythm. 2019;16(6):872-879. doi:10.1016/j.hrthm.2019.01.008.