Authors: Quinton Nannet, MD, (Emergency Medicine Resident, Atrium Health’s Carolinas Medical Center), Christine Murphy, MD (Emergency Medicine Attending, Medical Toxicologist, Atrium Health’s Carolinas Medical Center) // Reviewed by: James Dazhe Cao, MD (@JamesCaoMD, Associate Professor of EM, Medical Toxicology, UT Southwestern Medical Center, Dallas, TX); Alex Koyfman, MD (@EMHighAK); and Brit Long, MD (@long_brit)

Case:
A 34-year-old male presents to the emergency department (ED) via emergency medical services (EMS) for near syncope and altered mental status. The patient was picked up at a popular night club where his friends note he had been sniffing amyl nitrate “poppers.” EMS calls out that he is somnolent with non-bloody emesis on his shirt. The EMS crew notes the patient is hypoxic to the 80s and looks blue.
Initial ED vitals were HR 110; BP 65/48; RR 24 breaths/min; O2 sat 85% on 15L non-rebreather; Temp 37.8 °C. Physical exam reveals a patient who can speak with clear lung sounds, 2+ pulses, and a GCS of 15 but with marked cyanosis and dusky skin.
When drawing labs, the patient’s blood is appreciably dark. Chest x-ray and EKG are unremarkable. His venous blood gas demonstrates a pH of 7.41, pCO2 38, pO2 105, and methemoglobin of 35%.
Questions:
- What is the cause and mechanism of methemoglobinemia?
- How does methylene blue treat methemoglobinemia?
- How can methylene blue be used as a vasopressor in the ED?
- What are some current alternative uses of methylene blue?
Background: Methemoglobinemia1
- Methemoglobinemia occurs when the iron molecule in hemoglobin (Hgb) becomes oxidized from the ferrous (Fe2+) to ferric (Fe3+) state preventing it from being able to bind oxygen
- This resultant oxidation shifts the oxygen dissociation curve to the left, impairing oxygen distribution reducing the amount of functional hemoglobin
- Subsequently, patients with methemoglobinemia present with cyanosis, somnolence, dyspnea, and tachycardia
- As methemoglobin levels rise, the oxidative stress can also lead to hemolysis; particularly when reducing agents such as glutathione cannot be repleted, such as in glucose-6-phosphate-dehydrogenase (G6PD) deficiency
- The key diagnostic features include:
- Refractory pulse oximetry suggesting hypoxemia as the sPO2 will often remain < 85% despite 100% FiO2
- Cyanosis occurs despite an O2 saturation of ~ 80%, whereas cyanosis normally would occur < 75%
- Brown blood, as it cannot absorb oxygen and return to a normal red hue
- Causes of methemoglobinemia include anything that causes increased oxidative stress:


Figure 1. Methemoglobin-inducing agents. Image adapted from The Internet Book of Critical Care 2
Methylene Blue to Treat Methemoglobinemia
- Mechanism of Action:
- Methylene blue acts as a carrier for electrons to aid in reduction of ferric iron (Fe3+) back to ferrous iron (Fe2+)
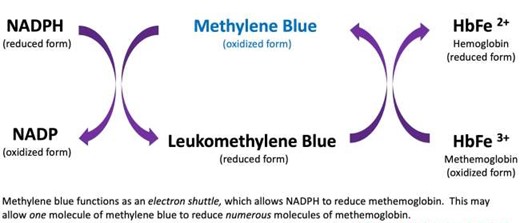
Figure 2. Methylene blue redox cycling. Image courtesy of The Internet Book of Critical Care (IBCC)2
- Indications:
- Symptomatic methemoglobinemia
- Neurologic: dizziness, headache, syncope, lethargy, coma, seizures
- Cardiovascular: dyspnea, exercise intolerance, fatigue, dysrhythmia, acidosis
- Methemoglobin level >30%
- Of note, cyanosis as a lone symptom is not an indication for treatment, as skin discoloration can be seen at methemoglobin levels of 3-20%
- Relative Contraindications:
- G6PD deficiency
- G6PD reduces NADP to NADPH
- NADPH in turn repletes oxidized glutathione stores in cells to limit free radical damage
- The G6PD pathway is the only source of glutathione in red blood cells making G6PD deficient patients more susceptible to oxidative stress which can lead to hemolysis
- NADPH is also needed to convert methylene blue to leukomethylene blue, which is the form that reduces methemoglobin (Fe3+) to hemoglobin (Fe2+), as illustrated in the image above. Thus, if NADPH stores are depleted due to ongoing oxidative stress, methylene blue is theoretically less beneficial
- Additionally, since methylene blue is an oxidizing agent, high doses of methylene blue could precipitate hemolysis through further oxidative stress
- Given the variability in G6PD activity, even in patients with “deficiency”, it is reasonable to consider treating with a small dose of methylene blue in patients with severe methemoglobinemia
- Alternative treatment options in patients with G6PD deficiency include exchange transfusions and hyperbaric oxygen3
- Ascorbic acid, frequently touted as a treatment for methemoglobin, can cause hemolysis in G6PD deficient patients and should not be used
- Methylene blue and serotonergic medications
- Methylene blue acts as a MAO-A inhibitor, meaning it blocks the degradation of amines (serotonin, norepinephrine, and dopamine)4
- When given in high doses at one time or over an extended duration methylene blue can precipitate serotonin syndrome
- This typically occurs in patients taking other medications with serotonin reuptake inhibition or those who have not had a long enough washout period after discontinuing a serotonin reuptake inhibitor5
- In most case reports noting this effect, patients received > 7 mg/kg. However, if a patient is on multiple medications blocking serotonin reuptake or increasing serotonin in the synapse, this could happen at lower doses.
- Again, this is a relative contraindication, as the risks of shock may outweigh the risks of serotonin syndrome
- Dosing3:
- 1-2 mg/kg IV over 5 minutes with a max initial dose of 100 mg for pediatric and adult patients
- For neonates, smaller doses of 0.3 mg/kg to 1 mg/kg are effective
- Most patients should improve clinically within minutes, and methemoglobin levels should reduce to < 10% within an hour
- If symptoms still persistent after 60 minutes, an additional 1 mg/kg can be given
- Failure to respond to two doses of 1 mg/kg should raise questions of G6PD deficiency or an alternative diagnosis
- Repeat dosing may be required if there is continued exposure to the cause of the patient’s methemoglobinemia, i.e., the drug continues to be absorbed or has a slow elimination time like dapsone
- Side effects:
- Transiently and falsely lowered pulse oximetry readings during administration
- Blue-green urine discoloration
- Infusion site discomfort
- The most common side effects noted in Goldfrank’s by healthy individuals receiving doses > 2 mg/kg were: extremity and back pain, chromaturia, dizziness, diaphoresis, nausea, skin discoloration, and headache3
- Doses > 7 mg/kg are typically thought as an upper limit and can potentiate further methemoglobinemia
- G6PD deficiency
- Symptomatic methemoglobinemia
Methylene Blue to Treat Vasoplegic Shock
- Mechanism of Action: Methylene blue causes vasoconstriction by inhibiting nitric oxide synthase and the conversion of guanine triphosphate to cGMP
- Indications: Distributive shock states
- Vasoplegia following cardiac surgery6, 7
- Sepsis8
- Dosing:
- Varies depending on sources and hospital practices:
- Bolus: 1-2 mg/kg IV
- If improvement noted, consider infusion from 0.5 – 2 mg/kg/hour
- Varies depending on sources and hospital practices:
Alternative Uses for Methylene Blue:
- Methylene Blue for Anaphylaxis9, 10

Figure 3. Methylene blue inhibits guanylate cycle and prevents vasodilatation. Image created by Dr. Nannet, adapted from Circulation Research11
-
- The pathophysiology of anaphylaxis is multifactorial, but it is known that the release of nitric oxide plays a large component
- The inhibition of guanylate cyclase by methylene blue reduces the vasodilatory effects of nitric oxide and can be considered in treating the hypotension associated with anaphylaxis

Figure 4. Electron transport chain in the mitochondria. Image courtesy of Fvasconcellos 22:35, 9 September 2007 (UTC), Public domain, via Wikimedia Commons
-
- One of proposed mechanisms of metformin toxicity is via inhibition of the mitochondrial respiratory chain complex I in the electron transport chain preventing aerobic metabolism and promoting anaerobic metabolism and lactic acidosis, (http://www.emdocs.net/toxcard-metformin-toxicity/)
- Methylene blue is theorized to act as a NADH electron acceptor, bypassing the electron transport chain when inhibited by metformin
Clinical Pearls
- Methemoglobinemia is the result of hemoglobin oxidation which prevents oxygen binding resulting in refractory hypoxemia, cyanosis, and brown or “chocolate” blood
- The treatment for methemoglobinemia is methylene blue which acts as an electron shuttle to reduce oxidized hemoglobin
- Methylene blue is relatively contraindicated in patients with G6PD deficiency due to the risk of hemolysis
- Methylene blue is dosed at 1-2 mg/kg IV over 5 minutes for methemoglobinemia, or you can give a bolus of 1-2 mg/kg and infusion of 0.5-2 mg/kg/hour for refractory vasoplegic shock
References
- Agarwal A.M., & Prchal J.T. (2015). Methemoglobinemia and other dyshemoglobinemias. Kaushansky K, & Lichtman M.A., & Prchal J.T., & Levi M.M., & Press O.W., & Burns L.J., & Caligiuri M(Eds.), Williams Hematology, 9e. McGraw Hill. https://accessmedicine.mhmedical.com/content.aspx?bookid=1581§ionid=101238113
- Farkas, J. (2021, November 30). Methemoglobinemia. EMCrit Project. Retrieved March 27, 2022, from https://emcrit.org/ibcc/methemoglobinemia/
- Hoffman RS, Nelson LS, Goldfrank LR, Howland MA, Lewin NA, Smith SW. Goldfrank’s Toxicologic Emergencies, Eleventh Edition. McGraw-Hill Education; 2019.
- Ramsay, R. R., Dunford, C., & Gillman, P. K. (2007). Methylene blue and serotonin toxicity: inhibition of monoamine oxidase A (MAO A) confirms a theoretical prediction. British journal of pharmacology, 152(6), 946–951. https://doi.org/10.1038/sj.bjp.0707430
- Ng BK, Cameron AJ. The role of methylene blue in serotonin syndrome: a systematic review. Psychosomatics. 2010 May-Jun;51(3):194-200. doi: 10.1176/appi.psy.51.3.194. PMID: 20484716.
- Levin R, Degrange M, Bruno G, et al. Methylene blue reduces mortality and morbidity in vasoplegic patients after cardiac surgery.Ann Thorac Surg. 2004;77(2):496-499. https://www.ncbi.nlm.nih.gov/pubmed/14759425.
- Mehaffey JH, Johnston LE, Hawkins RB, Charles EJ, Yarboro L, Kern JA, Ailawadi G, Kron IL, Ghanta RK. Methylene Blue for Vasoplegic Syndrome After Cardiac Operation: Early Administration Improves Survival. Ann Thorac Surg. 2017 Jul;104(1):36-41. doi: 10.1016/j.athoracsur.2017.02.057. Epub 2017 May 24. PMID: 28551045; PMCID: PMC5523819.
- Kwok ES, Howes D. Use of methylene blue in sepsis: a systematic review. J Intensive Care Med. 2006 Nov-Dec;21(6):359-63. doi: 10.1177/0885066606290671. PMID: 17095500.
- Evora PR, Simon MR. Role of nitric oxide production in anaphylaxis and its relevance for the treatment of anaphylactic hypotension with methylene blue. Ann Allergy Asthma Immunol. 2007 Oct;99(4):306-13. doi: 10.1016/S1081-1206(10)60545-5. PMID: 17941276.
- Rodrigues JM, Pazin Filho A, Rodrigues AJ, Vicente WV, Evora PR. Methylene blue for clinical anaphylaxis treatment: a case report. Sao Paulo Med J. 2007 Jan 4;125(1):60-2. doi: 10.1590/s1516-31802007000100012. PMID: 17505688.
- Friebe A, Koesling D. Regulation of nitric oxide-sensitive guanylyl cyclase. Circ Res. 2003 Jul 25;93(2):96-105. doi: 10.1161/01.RES.0000082524.34487.31. PMID: 12881475.
- Warrick BJ, Tataru AP, Smolinske S. A systematic analysis of methylene blue for drug-induced shock. Clin Toxicol (Phila). 2016 Aug;54(7):547-55. doi: 10.1080/15563650.2016.1180390. Epub 2016 May 19. PMID: 27196698.
- Graham RE, Cartner M, Winearls J. A severe case of vasoplegic shock following metformin overdose successfully treated with methylene blue as a last line therapy. BMJ Case Rep. 2015 Jul 6;2015:bcr2015210229. doi: 10.1136/bcr-2015-210229. PMID: 26150642; PMCID: PMC4493209.






1 thought on “ToxCard: Methylene Blue”
Pingback: Quiz 173, December 30th, 2022 – The FOAMed Quiz